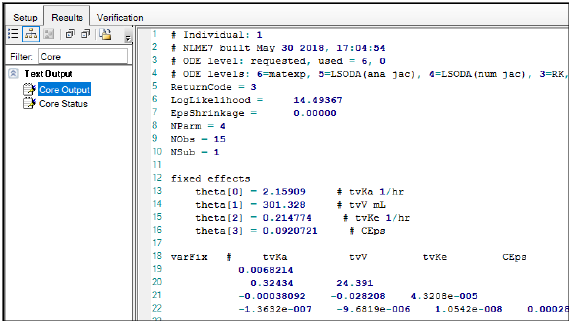
The advantages of the IPL model over in vivo inhalation studies is that the lung-delivered dose can be closely controlled without passing the nose and the lung deposited dose can be measured directly by mass balance calculations ( 6, 7). The isolated perfused rat lung (IPL) model is an ex vivo absorption model that allows parameters like epithelial permeability, solubility in epithelial lining fluid (ELF), dissolution rate, and particle wetting as well as tissue retention to be determined by computational analysis ( 4, 5). To ensure the efficient and successful development of inhalation drug products, improved knowledge about the pulmonary drug absorption, i.e., dissolution, permeability, and tissue retention of the API in the lungs, is needed ( 3). These properties enhance the pharmacological effect and decrease the dosing frequency, which improves compliance and reduces the risk of systemically adverse effects ( 2). Optimal pulmonary drug delivery of locally acting active pharmaceutical ingredients (APIs) includes high local concentration, extended lung residence time, and low systemic concentration ( 2). Pulmonary drug delivery is the preferred administration route for the treatment of lung diseases such as asthma, chronic obstructive lung disease, and cystic fibrosis ( 1). It could therefore be advantageous to base predictions of drug performance on IPL data rather than on in vitro data during drug development to increase mechanistic understanding of pulmonary drug absorption and to better understand how different substance properties and formulations might affect in vivo behavior of inhalation compounds. Furthermore, simulations using ex vivo input parameters performed significantly better than simulations using in vitro input parameters in predicting in vivo lung absorption. Simulations using ex vivo input parameters were within twofold average difference (AAFE < 2) from experimental in vivo data for all compounds except one. The performance of simulations using ex vivo input parameters was compared with simulations using in vitro input parameters, to determine whether and to what extent predictability could be improved by using input parameters determined from the more complex ex vivo model. Absorption parameters determined from IPL data ( ex vivo input parameters) in combination with intravenously determined pharmacokinetic data were used in a biopharmaceutics model to predict experimental rat in vivo plasma concentration-time profiles and lung amount after inhalation of five different inhalation compounds. This study aims to investigate the potential use of IPL data to predict rat in vivo lung absorption. The ex vivo isolated perfused rat lung (IPL) model has been demonstrated to be a useful tool during drug development for studying pulmonary drug absorption.
